Structural Optimization
Computational Modeling and Simulation
Structural optimization, structural analysis, computational fluid dynamics, dynamic analysis, design for manufacturing, and material molding may be necessary for your device. Our computational modeling and simulation services can help reduce timelines and decrease costs as you meet your project goals.
Topology/Shape Optimization
Structural optimization is an important tool in the design process. The methods topology and shape optimization can be used to optimize the geometry of a device early in the product development process which can significantly reduce timelines. Reducing timelines with structural optimization will decease your costs significantly.
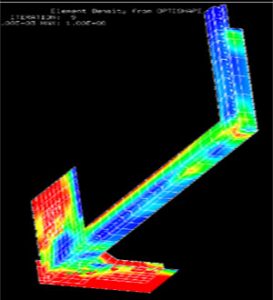
Topology Optimization (Red area requires higher stiffness)
Structural Analysis (Linear and Nonlinear)
Linear structural analysis can be utilized for the design and performance evaluation of many medical devices. Certain medical devices such as balloon expandable and self-expanding (nitinol) stents, however, operate beyond the linear elastic range – i.e., plastic, superelastic, and hyperelastic. We can perform structural analysis of the devices with highly nonlinear materials. Also our experts have extensive experience with analyses involving geometrical nonlinearity such as contact between device components and between device and body parts (blood vessels and other).
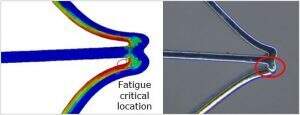
Nonlinear analysis: fatigue life evaluation of a stent
Computational Fluid Dynamics and Fluid Structure Interaction
Computational fluid dynamics (CFD) and fluid structure interaction (FSI) are utilized to assess the impact of blood flow or other types of body fluid on medical implants. We can leverage CFD and FSI to optimize device design, understand device performance, and troubleshoot device failures.

Fluid Flow analysis of a valve
Dynamic Analysis
When there is time dependent movement of a device such as a guidewire tracking, dynamic analysis can be utilized for evaluating performance and optimum design of products.
Design for Manufacturing
Considering manufacturability of products during the device design stage is very important for avoiding significant timeline delays in the later stages of development.
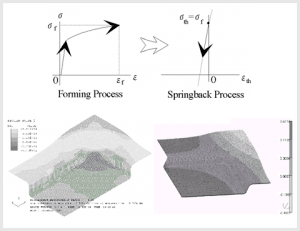
Deep Drawing and Springback of a Rectangular Pan
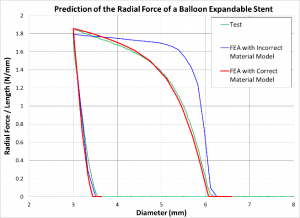
Material Modeling of Biocomatible Materials
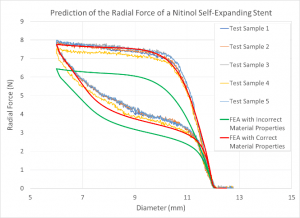
Together with highly sophisticated material testing techniques, we can create models of biocompatible materials such as nitinol, stainless steel, cobalt chrome, titanium, and polymers which are a key component to achieve accurate results in finite element analysis.
Influence of Material Properties on the Analysis Results:
OUR COMMITMENT
We are committed to consistently performing services with high quality, that deliver exceptional results, and add value to the client’s business.
For client surveys sent since 2024, we received ratings of 4.99/5 (16).
